+91 755 669 1338
+91 755 669 1338
 abhijit@iiserb.ac.in
abhijit@iiserb.ac.in
Fluorescent materials plays an important role in scientific research owing to their potential applications in the fields of sensors, cellular imaging and optoelectronic devices. Environment sensitive fluorescent molecules with high photostability and quantum yield are of increasing demand. Most of the conventional organic dyes, like perylene, pyrene etc. are non-fluorescent in the aggregated or solid state, which limits their usage in practical applications. The decrease of fluorescence intensity is due to the π-π stacking induced by molecular aggregation. Fine tuning of molecular interactions may lead to the development of novel fluorescent materials with strong emission in the aggregated (nanoparticles) and solid-state. Molecular motions (free rotation of specific bonds/ units) in solution enhance the non-radiative decay channels leading to decrease in emission. However, in the aggregated or solid-state, the intramolecular motions are restricted due to the crystal packing forces, which block the non-radiative pathways, leading to enhanced fluorescence.
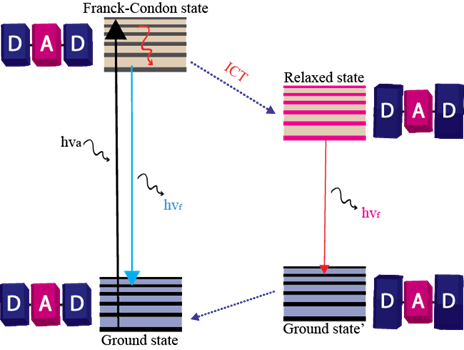
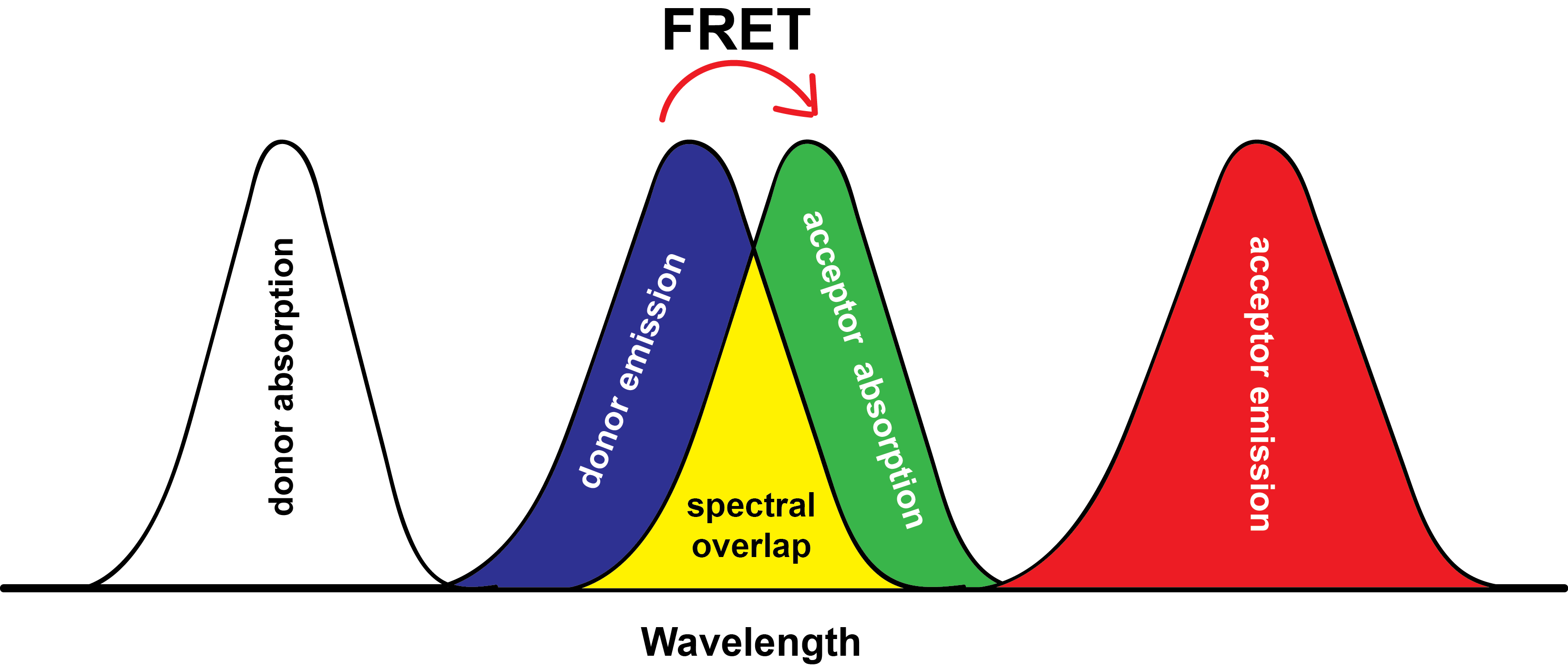
Apart from strong fluorescence, it is desirable if the probe molecules exhibit far-red or near-infrared (FR/NIR) emission. FR/NIR emission is advantageous in fluorescence imaging as it benefits from high penetration depth and low biological autofluorescence interference. The introduction of donor−acceptor (D-A) type molecular structures is one of the most prominent approaches to achieve lower energy absorption and emission. The intramolecular donor-acceptor interaction allows tuning of the energy difference between the highest occupied molecular orbitals (HOMO) and the lowest unoccupied molecular orbitals (LUMO). Hence, the tuning of HOMO-LUMO energy gap (band gap, ΔE) is an important approach to develop molecules/ materials with FR/NIR emission.
Scheme: Donor-Acceptor molecules
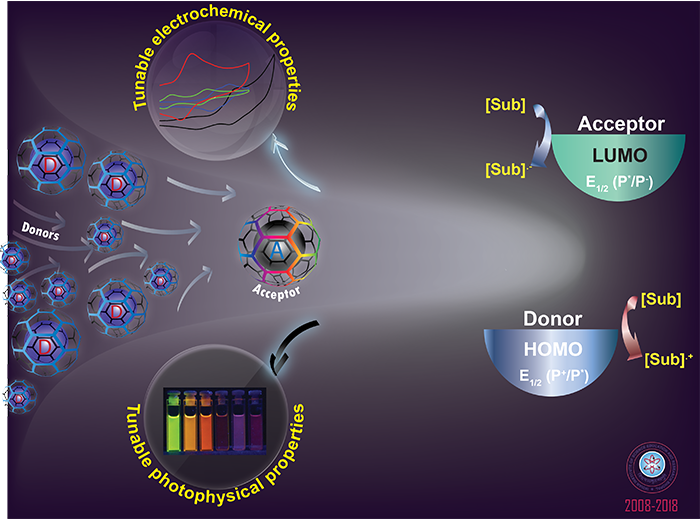
Tuning of fluorescence by Förster resonance energy transfer (FRET)